Hydrogen fuel cells hold great promise for future automotive transportation because of their higher overall energy efficiency and potential zero carbon emissions when compared to internal combustion engines. However, the use of costly platinum (and other precious metals) for accelerating the sluggish oxygen reduction reaction (ORR) in proton exchange membrane fuel cells (PEMFCs) has precluded their widespread deployment for electric vehicle applications. Although extensive research efforts have been devoted to minimizing Pt usage and enhancing its intrinsic activity via alloying and nanostructuring , replacing Pt with nonprecious metals or oxides represents a more promising strategy. Compared to PEMFCs in which the catalyst is estimated to contribute about 40% of the fuel cell stack cost, the primary advantage of anion exchange membrane fuel cells (AEMFCs) is that they enable the use of nonprecious transition metal–based ORR catalysts because of the improved catalyst stability in alkaline electrolytes. In an effort to facilitate the ORR kinetics in alkaline medium, a broad spectrum of nonprecious catalysts has been extensively studied, including metal-nitrogen-carbon, transition metal oxides, and perovskites. In particular, transition metal oxides, especially Co-Mn spinel oxides, have exhibited a power density of over 1 W cm−2 in membrane electrode assemblies (MEAs). Unfortunately, the low intrinsic electrical conductivity of those semiconducting spinel oxides has prevented further improvements in their ORR activity. An ideal ORR electrocatalyst should have an active surface responsible for catalyzing the ORR process and a conductive bulk to facilitate the charge transfer. Thus, developing conductive nonprecious catalysts represents a viable and attractive approach to circumvent the conductivity challenges and enhance ORR performance.
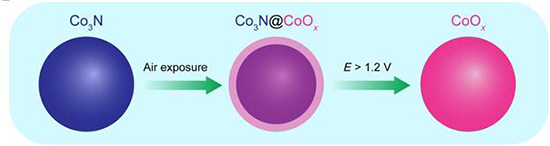
The research team reported on a group of nonprecious TMNs as potential ORR catalysts in alkaline medium. They first synthesized a family of carbon-supported metal nitrides via a facile nitridation strategy and systematically studied their morphologies and structures. Using surface and elemental characterization tools, they established that all the synthesized nitrides adopt a nitride-core and oxide-shell configuration upon exposure to the atmosphere. Electrochemical measurements in alkaline electrolyte showed that Co3N/C, MnN/C, and Fe3N/C displayed promising ORR activity, with Co3N/C exhibiting the highest ORR performance, comparable to commercial Pt/C. They then demonstrate that a peak power density of 700 mW cm−2 can be achieved with Co3N/C as cathode catalyst in an AEMFC, representing the highest MEA performance, among reported nitride cathode catalysts. Operando x-ray absorption spectroscopy suggested that while Co3N/C remains stable at potentials below 1.0 V, versus the reversible hydrogen electrode, it undergoes marked oxidation at more positive potentials. The new work may offer some insights into the design and development of active and durable TMNs as electrocatalysts for alkaline fuel cells and other energy systems and technologies. The authors led by Héctor D. Abruña, the Émile M. Chamot Professor in the Department of Chemistry and Chemical Biology in the College of Arts and Sciences published the findings in the journal Science Advances.
As long as combustion engines rule the streets and fill the skies with smog, it is hard to imagine a sustainable future for transportation. Hydrogen fuel cells, which convert hydrogen directly into electricity with only water and a small amount of heat as byproducts, are promising renewable alternatives.
Lately, however, more forgiving alkaline fuel cells have gained prominence, raising the possibility that less expensive metals, once ruled out for their vulnerability to acidic environments, might replace platinum in these gentler, next-generation fuel cells. The research team set out to engineer an inexpensive material, fit for an alkaline fuel cell, that would conduct electricity and catalyze the ORR reaction just as efficiently as platinum. Transition metal nitrides (TMNs) were an obvious choice. A class of compounds derived from cobalt, manganese, iron and other transition metals, TMNs conduct electricity and, when exposed to air, tend to form a thin oxygen-based outer shell that provides a perfect surface for catalyzing chemical reactions. After synthesizing a family of TMNs with conductive nitride cores and reactive oxide shells, the team tested each candidate catalyst in a model hydrogen fuel cell.
In summary Cornell chemists have discovered a class of nonprecious metal derivatives that can catalyze fuel cell reactions about as well as platinum, at a fraction of the cost. This finding brings closer a future where hydrogen fuel cells efficiently power cars, generators and even spacecraft with minimal greenhouse gas emissions. Those savings may help finally brings hydrogen fuel cells out of the laboratory and into the mainstream. If affordable, fuel cells could replace combustion engines and car batteries with a sustainable alternative that, fed a steady diet of hydrogen, never needs to recharge and wastes as little as 10% of the energy that goes into making it run. By comparison, a typical car engine wastes about 75% of its energy.

Emile M. Chamot Professor
Cornell University
The Abruña Group focuses on the development and characterization of new materials using a wide variety of techniques for fuel cells, batteries, and molecular assemblies for molecular electronics.
Reference
Zeng R, Yang Y, Feng X, Li H, Gibbs LM, DiSalvo FJ, Abruña HD. Nonprecious transition metal nitrides as efficient oxygen reduction electrocatalysts for alkaline fuel cells. Sci Adv. 2022 Feb 4;8(5):eabj1584. doi: 10.1126/sciadv.abj1584.




