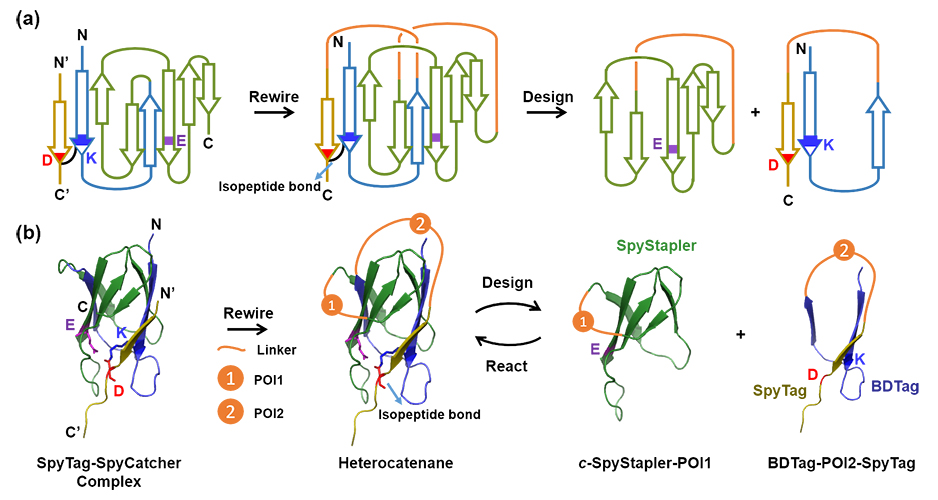
The emerging field of protein topology engineering is characterized by creating topologically nontrivial proteins with more functional benefits than their linear counterparts. Since the first preparation of artificial peptide catenanes, rotaxanes, and heterocatenanes through solid-phase peptide synthesis, this field has attracted significant research interest. For over a decade, researchers have been trying to make artificial topological proteins beyond simple linear or circular forms. According to past reviews, cellular synthesis of protein catenanes have been successfully achieved by emulating the essence of topological design in Nature using an entangling dimerization motif p53 and a genetically encoded SpyTag-SpyCatcher reaction to lock in the geometry. However, this approach is limited to homocatenanes involving two identical rings and the synthesis of programmable, complex protein heterocatenanes remains a big challenge. To address this, the structural complexity and topological diversity of protein architectures should be explored further.
Inspired by the insights of their previous research work, Xiao-Di Da (PhD candidate) and Professor Wen-Bin Zhang from Peking University recently developed a biologically enabled active template for the concise synthesis of protein heterocatenanes both in vivo and in vitro. This approach was based on the modification of the SpyTag-Catcher complex to initiate rationally designed and desirable change entanglement while retaining the catalytic activity for isopeptide formation to form the mechanical bond. Their work is published in the journal, Angewandte Chemie.
Briefly, the method was developed by rewiring three individual fragments in the SpyTag-SpyCatcher complex in three-dimensional space to induce programmable chain entanglement. Upon reconstitution, the fragment containing BDTag and SpyTag “threads” through the cyclic SpyStapler fragment which catalyzes the subsequent “clicking” reaction between BDTag and SpyTag to accomplish the cyclization and form the entangled heterocatenane structure through isopeptide bonding. The general feasibility of the heterocatenanes formation approach was validated by inserting various proteins of interest into the gene to make various protein heterocatenanes modularly. The properties of these protein heterocatenanes were further tested by evaluating the target proteins’ stability, thermal and freeze-thaw properties.
As proof of the catenane topology, the authors performed orthogonal proteolytic digestion experiments using two mutually orthogonal proteolytic sites designed on each of the two precursors. When treated with only one of the proteases, the heterocatenane generated two different combination of products; when treated by the two proteases together, it generated another different combination of products. The reported digestion results agreed well with the expectations thus demonstrating the feasibility of the catenane topology.
The reaction proved efficient for forming heterocatenanes with precisely controlled ring sizes and broad structural diversity. It was worth noting that the method allowed for the insertion of various model proteins, such as elastin-like polypeptides, fluorescent proteins, and dihydrofolate reductase, in the linker regions implying its feasibility in forming heterocatenanes from both unstructured and folded protein precursors. This was attributed to the fact that the strategy is autonomous and fully genetically programmable. Other than providing efficient means for bioconjugation for integrating multiple functions, such heterocatenation were also observed to exert little influence on the three-dimensional structures of the folded proteins, preserving their native properties while enhancing their stabilities against proteolytic digestion, thermal inactivation as well as freeze-thaw induced mechanical denaturation.
In addition, not only the efficiency of this active template may be further improved by directed evolution, other covalent-bond-forming protein domains could also be used to develop similarly effective, mutually orthogonal active templates for creating unconventional protein topologies. “I am very excited about the method.” Professor Zhang says, “It means that highly complex topologies such as protein Olympic rings may be constructed in as few as one step from the corresponding component protein precursors!”
In summary, the study by Ms Xiao-Di Da and Professor Wen-Bin Zhang demonstrated a new active template for the synthesis of protein heterocatenanes both within and outside living organisms. Based on the presented results, the strategy provides a “threading-and-clicking” tool for designing protein architecture in topology. The same sentiments were echoed by Professor Zhang who noted that the study provides new and useful insights that would provide a versatile path for advancement in protein-topology engineering.

Xiao-Di Da received bachelor degree in chemistry from Nankai University in 2015, and then went to Peking University to pursue a PhD degree in polymer science under the supervision of Prof. Wen-Bin Zhang. Her PhD work mainly focus on protein topology engineering including the synthesis of various topological protein architectures and the structure-property relationships. She developed a concise and modular “active template” synthesis of protein heterocatenanes.

Wen-Bin Zhang is currently an Associate Professor at the College of Chemistry and Molecular Engineering of Peking University. He received his BS from Peking University in 2004 and his PhD in Polymer Science from the University of Akron in 2010. He continued there as a postdoc for one year, before moving to Caltech for a second postdoc. He started his independent career at Peking University in 2013 and was promoted to a tenured associate professor in 2019.
His research in the past decade has been the pursuit of precision in macromolecular science at both molecular and supramolecular level. His goal is to integrate the design principles and building blocks of both synthetic and biological polymers for the development of precision macromolecules with unique functions for health-related applications. He received the “1000 Talent Award (Youth)” in 2013, the “Distinguished Lectureship Award” from the Chemical Society of Japan in 2017, and the Distinguished Young Scholar Award from NSFC in 2019.
Reference
Da, X., & Zhang, W. (2019). Active Template Synthesis of Protein Heterocatenanes. Angewandte Chemie, 131(32), 11214-11221.